100% Private and Confidential
Experienced & Specialized Care Team
24/7 Resources Accessible From Any Device
Personalized, Clinically-Validated Activities
Trusted by Health Plans and Leading Employers
Ready When You Need Us
ALAViDA Substance Use, a product of LifeSpeak Inc., provides easy access to substance use care: wherever you are, whenever you need it. You set your goals. We help you meet them.
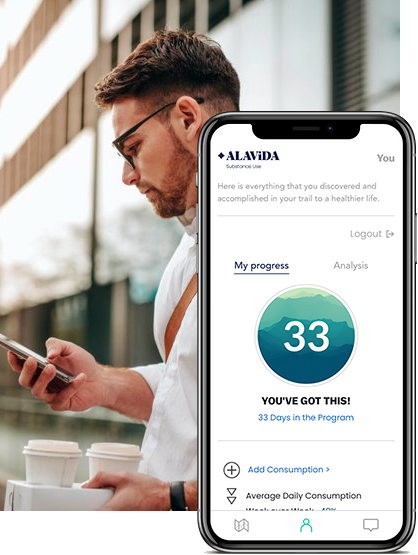
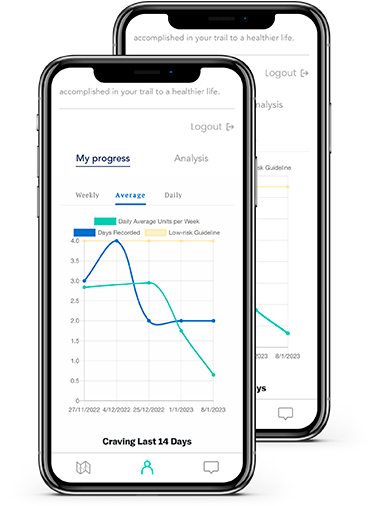
Virtual Companion
An immersive experience developed by medical professionals and delivered by technology.
Dedicated Care Team
A team of proactive care professionals just for you. Set your goals. Let us help.
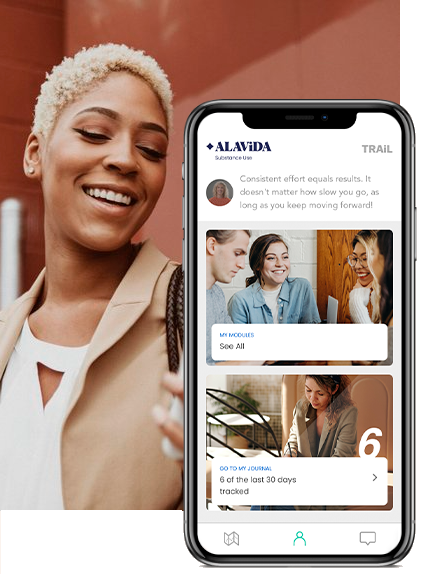
Moderated peer support
Optional group sessions where you can feel comfortable and safe as you connect with others going through similar experiences
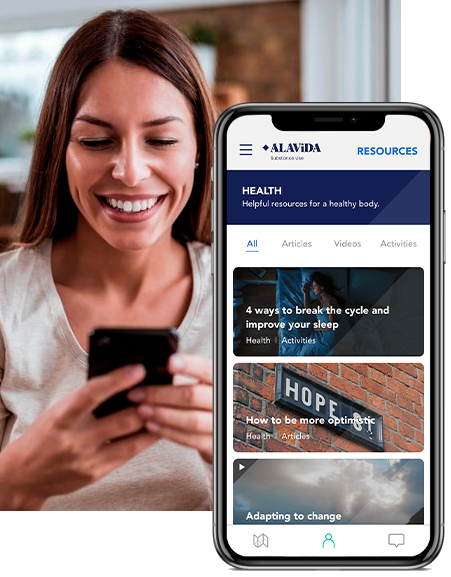
24/7 Resources
Support is always within reach. Our solution also connects you with your personalized care team to help keep you on track.
iCBT
Evidence-based therapeutic approaches to help you understand your substance use, strengthen decision-making, and take control.
Awareness Tools
Learn more about substance use and management; access support and put yourself first.
Any Substance
Our team specializes in substance use. We can help with anything you’re struggling with.
Anytime
We offer immediate access to an array of helpful resources, available 24/7.
Anywhere
Right from your phone. Get private and confidential access wherever you are.
Ready to add ALAViDA to your benefits?
10% of employees will experience challenges with substance use, but only 1 in 10 will access treatment. It’s time to change that!